The European Medicines Agency (EMA) issued an update on the safety concerns for Actos on October 21, indicating that the drug is still a valid diabetes treatment. But, due to the Actos bladder cancer risk, it should only be used when other treatments have failed. This update is similar to the latest one issued by United States regulators.
Dr Shezad Malik Law Firm has offices based in Fort Worth and Dallas and represents people who have suffered catastrophic and serious personal injuries including wrongful death, caused by the negligence or recklessness of others. We specialize in Personal Injury trial litigation and focus our energy and efforts on those we represent.
Articles Posted in Medical News
J&J Vaginal Mesh Approved by FDA Based on Recalled Device
As a Johnson & Johnson Gynecare TVT Trans Vaginal Mesh injury attorney and Texas medical doctor, I am providing this timely update.
Johnson & Johnson (JNJ), the world’s second-biggest health-care products maker, is facing many lawsuits over its Gynecare TVT vaginal implant. The Gynecare TVT implant is based on a similar device pulled from the medical device market more than a decade ago for safety reasons.
The lawsuits are the latest to implicate the shoddy and perfunctory approval process for medical devices at the U.S. Food and Drug Administration, which has cleared faulty hip implants and malfunctioning defibrillators.
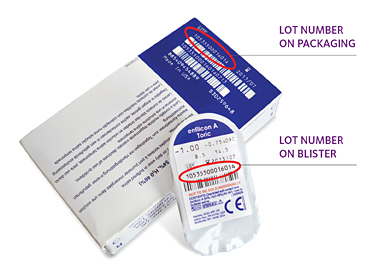
CooperVision Contact Lens Recall Class 1 Status
According to the FDA, silicone oil is the cause of problems with Aviara contact lenses sold by CooperVision, which were removed from the market in August after a number of consumers suffered torn corneas, hazy vision and other potentially serious complications. Nearly two months after problems were first announced, contact lens giant CooperVision Inc. and federal health regulators have amped up public warnings about nearly 780,000 recalled contact lenses, urging users to return the potentially defective products and to seek medical care if they show symptoms of harm.
The FDA announced on Friday that it was classifying the CooperVision Aviara contact lens recall as a Class 1 medical device recall, which is the most serious category the agency can use to label a recall. The class 1 designation means that the FDA believes that use of a device carries a reasonable probability of serious adverse health consequences or death.
More than 778,000 CooperVision Aviara contact lenses were impacted by the recall, which was not widely publicized until the FDA criticized the company last week for failing to take sufficient steps to make sure consumers were aware of the contact lens problems.
Yaz Birth Control Coma and Blindness
As a Yaz Birth Control Coma and Blindness Injury attorney and Texas medical doctor I am providing this YAZ, Yasmin and Ocella update.
ABC’s chief law and justice correspondent, Chris Cuomo, interviews Carissa Ubersox, a former pediatric nurse who started taking Bayer’s Yaz birth control pills when she wanted to look her best for her wedding and “saw commercials suggesting help with bloating and acne.” Two months after Ubersox started to take Yaz birth control pills, she developed massive blood clots in both lungs and fell into a coma that lasted for two weeks. When she woke up, she was blind.
All birth control pills come with some risk of developing blood clots, and Bayer “cites its own studies as proof that Yaz is just as safe as other birth control pills.” However, Dr. Susan Jick of the Boston University School of Medicine has authored an independent study involving a million women that finds Yaz’s risk to be two to three times higher than other birth control pills. ABC reports that two Bayer-sponsored studies find no difference in risk, while four independent studies all find increased risk.
Medtronic Infuse Side Effects and Lawsuits
As a Dallas Medtronic Infuse Lawsuit and side effect attorney and medical doctor I am providing this update regarding the problems associated withe Medtronic Infuse medical product.
Medtronic, a medical device company is in the news regarding reports about its synthetic bone growth product Infuse. In 2008, the U.S. Department of Justice began investigating whether Medtronic had been illegally promoting “off-label” use of Infuse.
The Spine Journal reported in its June 2011 edition concerns about Infuse and Medtronic’s research. In 2004, a small group of doctors wrote some research papers stating that Infuse did not cause any harm to patients. The doctors who wrote the article would eventually receive millions of dollars from Medtronic.
DePuy Pinnacle Hip Replacement MDL Lawsuits
Visit msnbc.com for breaking news, world news, and news about the economy
As a DePuy ASR and DePuy Pinnacle litigation attorney, I am providing this timely update.
More than 500 lawsuits have been filed in Northern District of Texas federal court over DePuy Pinnacle hip replacements problems. The plaintiffs have alleged that DePuy hips, caused individuals to experience severe pain and early failure of their artificial hip implant.
DePuy ASR MOM Hips in the News
As a DePuy ASR Hip Recall attorney I am providing this update. There is a problem with metal on metal artificial hip implant devices, as highlighted on the NBC Nightly News on October 4th 2011.
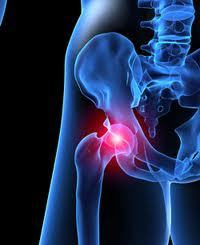
According to medical experts and bio-engineers, the DePuy ASR, and DePuy Pinnacle hip implants are failing, or have failed at an unacceptably high rate. In August of 2010, there was a recall on DePuy ASR hip implant device. The ASR DePuy hip implants were surgically implanted on tens of thousands of US citizens between 2005 and early 2010.
Symptoms of a recalled ASR DePuy hip implant failure include pain in the hip region, problems walking, swelling of the hip, or lack of flexibility in the area of the hip. NBC Nightly News with anchor Brian Williams, called attention to the DePuy hip implant recall, and the issues with metal on metal hip implants on Tuesday October 4th 2011 news segment.
DePuy Metal on Metal Hip and Metallosis
As a DePuy ASR and Pinnacle Replacement and Recall attorney I am providing this blog update.
As readers to my blog will note, I am a licensed Texas medical doctor as well as a product liability attorney. We are currently fielding many phone calls from concerned hip replacement patients and the biggest concern for them is residual hip pain post procedure.”Is that normal?” they ask. “How do we know if we are suffering from metallosis?” “My doctor is not concerned but I still feel that something is wrong?”
The answers to these questions remain elusive for several reasons. Firstly and foremost is the lack of research, disclosure and transparency from the hip manufacturers who knew or should have known about these problems and concerns.
Trans Vaginal Mesh TVM Lawsuits: The Next Big One?
According to the concerned women who are calling our offices; they believe that the FDA and other U.S. regulators have failed them by not requiring extensive testing before allowing Johnson & Johnson (JNJ) and other manufacturers to sell the type of surgical mesh implanted in them, to hold their pelvic organs in place. Now many these callers say that they cannot work, sleep through the night, or have sex with their partners due to endless pain.
There are about 270 lawsuits pending against J&J. In all, about 600 suits have been filed against it and other mesh makers, including C.R. Bard, Boston Scientific, and American Medical Systems, acquired in June by Endo Pharmaceuticals Holdings.
The U.S. Food and Drug Administration warned on July 13 of a fivefold increase in women suffering pain and injuries after surgeons inserted mesh through vaginal incisions. Pelvic Organ Prolapse (POP) occurs when the internal structures that support the pelvic organs such as the bladder, uterus and bowel, become so weak or stretched that the organs drop from their normal position and bulge or prolapse into the vagina. While not a life-threatening condition, women with POP often experience pelvic discomfort, disruption of their sexual, urinary, and defecatory functions, and an overall reduction in their quality of life.
Actos MDL Consolidation Over Bladder Cancer
As a Texas medical doctor and Actos Bladder Cancer Attorney I am providing this information and commentary. The maker of the world’s best-selling diabetes drug is facing many lawsuits as adverse medical research shows that taking the pill for more than a year raises the risk of bladder cancer.
A motion was filed asking for all federal Actos bladder cancer lawsuits to be centralized before one judge for coordinated handling during pretrial proceedings as part of an MDL, or multidistrict litigation.
There are about 12 lawsuits over Actos that have been filed in federal district courts. Experts believe that hundreds of complaints that will likely be filed on behalf of individuals who claim they developed bladder cancer from side effects of Actos.
 Dallas Fort Worth Injury Lawyer Blog
Dallas Fort Worth Injury Lawyer Blog