Dr Shezad Malik Law Firm is expanding their initiatives focused on identifying recipients of metal-on-metal hip implants, that has failed, or are in the process of failing.
While one specific type of metal-on-metal hip implant called the DePuy ASR has been recalled, the group expects there to be many more recalls of metal-on-metal hip implants, because of the high probability of failure.
The 2010 DePuy Hip Replacement Recall was instituted when DePuy Orthopaedics, Inc., a division of Johnson and Johnson. Johnson & Johnson recalled its ASR XL Acetabular metal-on-metal hip replacement system, on August 24, 2010.
According to the New York Times, “The so-called metal-on-metal hips, in which a device’s ball and joint are made of metal, are failing at high rates within a few years instead of lasting 15 years or more, as artificial joints normally do. The wear of metal parts against each other is generating debris that is damaging tissue and, in some cases, crippling patients.”
In order to make certain all metal-on-metal hip implant recipients, with a failed hip implant, get evaluated, the Dr Shezad Malik Law Firm wants to hear from any metal-on metal hip implant recipient, if their hip implant has already failed, or if the hip implant recipient is suffering the symptoms of a metal-on metal hip implant failure.
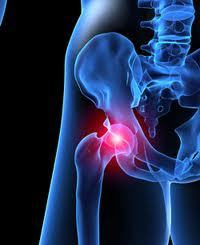
There are many symptoms, including pain, loss of flexibility, swelling, or loss of mobility.
One of the biggest problems we have with respect to our failed medical device, or recalled drug work, is the average US consumer never hears about a drug recall, or failures involving medical devices like metal-on-metal hip implants.
If you have a friend, or loved one, who is a recipient of a metal-on metal hip implant that has already failed, or is showing symptoms of a failure, such as swelling, pain in the region of the hip, loss of flexibility, or loss of mobility, please have them call us at 214-390-3189.
 Dallas Fort Worth Injury Lawyer Blog
Dallas Fort Worth Injury Lawyer Blog